The Critical Care Society of Southern Africa Consensus Guideline on ICU Triage and Rationing (ConICTri)
Â
 Background. In South Africa (SA), administrators and intensive care practitioners are faced with the challenge of resource scarcity as well as an increasing demand for intensive care unit (ICU) services. ICU services are expensive, and practitioners in low- to middle-income countries experience the consequences of limited resources daily. Critically limited resources necessitate that rationing and triage (prioritisation) decisions are routinely necessary in SA, particularly in the publicly funded health sector.Â
Purpose. The purpose of this guideline is to utilise the relevant recommendations of the associated consensus meeting document and other internationally accepted principles to develop a guideline to inform frontline triage policy and ensure the best utilisation of adult intensive care in SA, while maintaining the fair distribution of available resources.Â
Recommendations. An overall conceptual framework for the triage process was developed. The components of the framework were developed on the basis that patients should be admitted preferentially when the likely incremental medical benefit derived from ICU admission justifies admission. An estimate of likely resource use should also form part of the triage decision, with those patients requiring relatively less resources to achieve substantial benefit receiving priority for admission. Thus, the triage system should maximise the benefits obtained from ICU resources available for the community. Where possible, practical examples of what the consensus group agreed would be considered appropriate practice under specified South African circumstances were provided, to assist clinicians with practical decision-making. It must be stressed that this guideline is not intended to be prescriptive for individual hospital or regional practice, and hospitals and regions are encouraged to develop specified local guidelines with locally relevant examples. The guideline should be reviewed and revised if appropriate within 5 years.Â
Conclusion. In recognition of the absolute need to limit patient access to ICU because of the lack of sufficient intensive care resources in public hospitals, this guideline has been developed to guide policy-making and assist frontline triage decision-making in SA. This document is not a complete plan for quality practice, but rather a template to support frontline clinicians, guide administrators and inform the public regarding appropriate triage decision-making.Â
1. Introduction
The intensive care unit (ICU) provides a higher level of care than the general ward or intermediate care units, and is a place where
patients with potential or established organ failure can receive
close monitoring and life support treatment delivered by specially trained staff. [1] In SA, ICUs generally provide a high standard of intensive care; however, because of the expensive nature of intensive care services, there are a limited number of ICU beds and facilities available. [2-5] Rationing is therefore inevitable, and a priority system that is fair and efficient is required to ensure the ongoing provision of high-quality intensive care, with the best utilisation of available resources. Guidelines for making such ICU triage decisions are important to promote consistency, fairness and high standards of clinical decision-making at the bedside. [6-9] Transparently promulgated and communicated guidelines also promote effective communication with patients, their surrogates, the public and referring doctors.
The purpose of this guideline is to utilise the relevant recommendations of the associated consensus meeting document, [10] and other internationally accepted principles, to develop a guideline that informs frontline policy for patient triage, admission to and discharge from ICUs in SA. As stated in the Durban Declaration, all basic moral and ethical principles must be applied to ensure rational decision making in intensive care, but particularly because intensive care is a limited resource, that the social justice principle as a competing interest must be recognised. [11] The framework presented is designed to help maximise the use of ICU services to achieve the largest possible benefit for the most patients from available resources. This utilitarian ethical approach has been recommended by expert groups and ICU professional bodies internationally, [6,12-15] and was recommended by the accompanying South African triage consensus statement. [10]
In addition, and to ensure fairness, further principles that should be met are briefly summarised (Table 1). The broad principles provided should serve to assist individual ICUs to develop their own local policy that best suits the specific requirements of the hospital. Individual ICUs are therefore encouraged to further define their scope of practice, service provision, and develop detailed guidelines for the implementation of triage, provided that the patient population is served according to the above principles.
2. Methods
The framework arises from a detailed discussion during the afternoon session of a full day, face-to-face round-table meeting at the Critical Care Society of Southern Africa (CCSSA) National Congress held at Sun City on 18 October 2017, and was informed by findings of the accompanying consensus statement, [10] and previously described triage practices and available international consensus guidance. [6,15-20] It thus serves as an application tool to assist the practical application of the principles agreed at the consensus meeting. [21] After the round table meeting, a draft of the written guideline was circulated to the consensus group for comments and suggested modifications. After 2 formal rounds of consultation, the draft guideline was made openly available on the CCSSA website https://www.criticalcare.org.za/ConICTri/Whatis from August 2018 for 3 months, and both invited participants and CCSSA members were asked to review and comment on the proposed draft. The site was open to public view during this period. In response to suggestions received via the website portal, small additional changes were finalised during November 2018. Opinions received via the website were generally positively aligned with the content of the guideline. The guideline, with relevant open external consultation additions included, was circulated to all members of the consensus group in December 2018 for consensus and final approval.
3. Consensus guideline
The overall conceptual framework for the triage process is summarised in Fig. 1. The guideline text is divided into 7 parts to allow the components of the framework to be described in detail. Where possible, practical examples of what the consensus group agreed would be considered appropriate practice under qualified South African circumstances are provided to assist clinicians with practical decision-making. It must be stressed that this guideline is not intended to be prescriptive for individual hospital or regional practice, and hospitals and regions are encouraged to develop specified local guidelines with locally relevant examples.

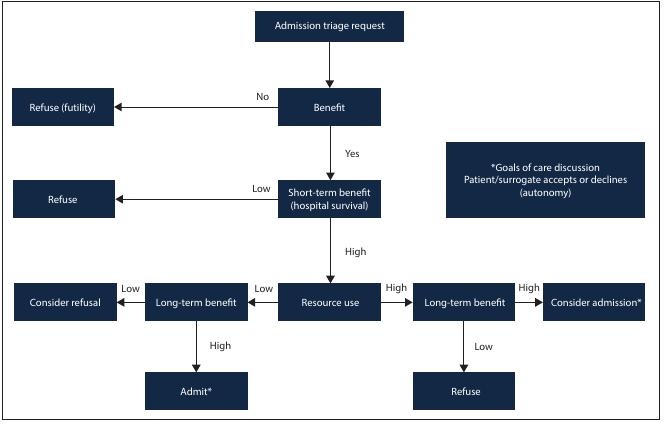
Fig. 1. A triage (prioritisation) decision is a complex clinical decision made when ICU beds are limited. A structured decision-making process is important to maximise transparency and improve consistency in decision-making. A clinical estimation of likely t (outcomes from ICU admission compared with outcomes expected if patient remained in the ward/other care area) is necessary, so that patients who will benefit most from ICU are given priority. Based on the expert group’s experience, a hypothetical example of an acceptable triage threshold for an ICU that routinely performs triage at least on a daily basis, would approximate at least a 20 - 30% chance of survival for a severely ill patient at 3 - 6 months (threshold for long-term t). is assumes a 5% chance of survival if the patient was at their original level of care (usually a general ward). Some examples of conditions that the expert group believe would fail to meet these criteria are provided (Table 2). In addition, some examples of conditions that it would meet the criteria for cial care or futility (short and long term) are provided (Table 5).
This conceptual algorithm outlines a recommended process for making an individual triage decision. Each decision is made on the basis of an agreed triage threshold for the particular setting (e.g. stricter thresholds may be required during the winter surge, and academic units may require special arrangements to support elective surgery). Long-term t should include an assessment of expected quality of life, if considered appropriate (Table 3). As these examples are hypothetical, each unit should develop individual policies that take the above framework into account, but with clinical content and thresholds that are ed for local requirements. (Figure adapted from Joynt and Gomersall. [20] )
*Before the final decision to admit to ICU, and if admission is to be ered, patient preference regarding desire for admission should be explored with the patient or the patient’s surrogate when appropriate.
4. Triage priority for admission
Patients categorised as high priority should be admitted to the ICU whenever possible (although in some units and regions of SA, the ICU resource shortage is so severe that not all high-priority cases can be admitted). If the ICU is fully occupied, attempts should be made to transfer these patients to other units within the region, if such a possibility exists. High-priority patients fall into the following broad categories:
1. Critically ill patients with acute organ failure/s who require life support therapies that can only be provided in the ICU and are likely to derive substantial incremental benefit from ICU care compared with alternatives outside the ICU such as general ward care. Such therapies include invasive ventilator support, continuous vasoactive drug infusions, continuous renal replacement therapy, extracorporeal membrane oxygenation, and other forms of advanced life support.
2. Critically ill patients who require intensive monitoring and potentially immediate interventions that can only be provided in an ICU. In comparison with monitoring and treatment available outside the ICU, such patients should also be expected to derive substantial benefit in terms of survival probability, quality and length of life.
Low-priority patients are critically ill patients who are likely to derive some, but not substantial, benefit from ICU care, and may be refused ICU admission when resources (adequately staffed and equipped ICU beds) are limited. Thus, the available ICU resources are preserved for use by high-priority patients.
The threshold for determining the triage decision (magnitude of benefit required to be considered substantial) will be determined primarily by the balance of local ICU provision and demand for ICU services. For example, in a well- resourced hospital, with few referrals for ICU admission, only patients with a small chance of incremental benefit may be categorised as low priority and need be refused admission. Conversely, in a severely under-resourced hospital with many referrals for ICU admission, patients with a much greater chance of benefit may find themselves refused admission as the queue for admission will be filled with patients with a very high likelihood of benefit. Thus, the determination and description of the triage threshold ultimately is made by the ICU management, after broad consultation with senior ICU personnel and, where necessary, other stakeholders.
In the interests of fairness and consistency, triage thresholds in individual units should be defined and openly documented as clearly as possible by the ICU management, and respected by those performing triage, as well as referring medical teams. It is recognised that the availability of ICU beds throughout the country is highly variable, [3,4] and consequently appropriate triage thresholds that are established for regions/clusters of units, or individual units, will vary, depending on the magnitude and consistency of pressure for beds. Triage thresholds may also vary from time to time, such as during a seasonal outbreak of respiratory or other infectious disease. [22,23] Under circumstances where two or more patients of high priority both require admission, and all cannot be admitted because the ICU is fully occupied, the patient/s with the highest priority should be admitted first; or, should these patients be judged as having equally high priority, then on a first-come, first-served basis.
It should not be forgotten that patients with an extremely good prognosis may also not necessarily derive substantial benefit from ICU care, compared with a lower level of care, and may be refused admission on the basis of triage, for example a patient with chronic obstructive pulmonary disease (COPD) responding to non-invasive ventilation in a ward would derive little extra benefit from ICU admission. It would then be reasonable to reserve the ICU bed for a more ill patient (perhaps with a worse prognosis), but one likely to derive greater incremental benefit from admission.

4.1. Decision-making process
Upon referral of a patient for possible admission to an ICU with limited capacity for admission, the triage (prioritisation) process identifies a spectrum of patients who will likely derive substantial incremental benefit from ICU admission, and therefore should receive priority for admission. It is acknowledged that triage is a complex clinical decision dependent on several factors. The use of scoring systems to objectively identify the triage threshold (sufficient incremental magnitude of mortality benefit likely to be derived from admission), while attractive, is not currently possible. [24-26] This question was discussed in more detail in the concurrent consensus statement. [10] Simple, accessible and rapidly available point of care predictive scoring systems for mortality, when available, are recommended to assist prognostication and therefore the clinical estimate of magnitude of likely benefit. Unfortunately, few are suited to use in the triage setting. [27,28] Thus, determining the triage threshold remains largely a considered clinical decision.
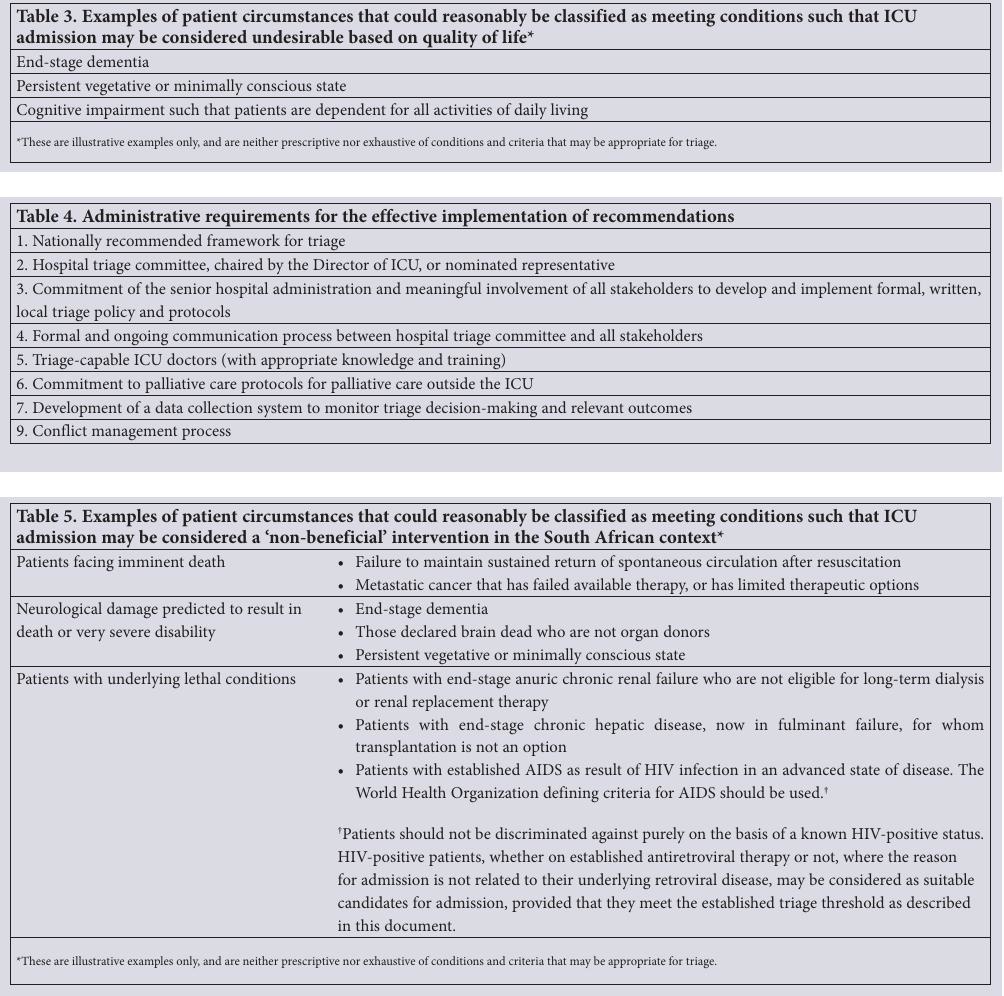
The algorithm in Fig. 1 describes how the clinical judgment of whether a patient meets the triage threshold should be framed, without prescribing the specific clinical requirements necessary to meet the triage threshold in individual units. To provide some practical guidance, an indicative example of criteria is provided in Tables 2, 3 and 5. Specific clinical requirements for individual units or groups of similar units, if deemed necessary, should be determined by local unit policy that will in turn be dependent on a local assessment of pressure for available beds. It is expected that these may be different from those provided in the examples that follow.
4.2. Setting a triage threshold
To assist individual units to develop and document processes and thresholds for triage decision-making based on local circumstances, some examples follow. Following the framework suggested in Fig. 1, the round-table participants constructed clinical descriptions or categories of patients that would not meet the triage threshold of ‘substantial benefit’ in a hypothetical unit with a daily requirement to refuse referred patients (Fig. 1, Tables 2, 3 and 5). In each of these clinical settings, the incremental difference in benefit from ICU admission would be expected to be small compared with that
achieved by general ward care alone. In all of these examples, the length of ICU stay was estimated to be relatively long, and thus be at least moderately costly in terms of expected ICU resource used. It must be stressed that these examples were constructed during and after the consensus meeting and are based on the experience of the clinical members of the consensus group. The triage thresholds set were based on a consensus of the members present, and are not intended to be prescriptive, but indicative of thresholds that the expert group considered to be at least reasonable in a unit with chronic resource limitations and the need to refuse referred patients on most days, because of insufficient available beds. During development of the examples, the severe burn criteria (as currently presented in Table 2) were considered to be too strict in one tertiary academic unit with relatively more resources, but considered too liberal to represent what was currently practised in a regional ICU situated in a smaller city with more limited resources. In addition, the greater expertise and clinical support in the tertiary unit made the odds of survival greater. These differences are expected, and provide an example of how local policy should adjust criteria to meet appropriate local resource conditions. For this reason, it is appropriate and important that one or more of the criteria in the examples provided here are adjusted, removed and/or others added to properly guide local practice. These adjustments, once incorporated into the local triage policy, should be reviewed and updated from time to time, as resource conditions may change.
For these examples, long-term benefit may reasonably be expected to fail to provide at least a 20 - 30% incremental chance of survival for a severely ill patient at 3 - 6 months to qualify for the triage threshold. This assumes no more than a 5% chance of survival if the patient was left at their original level of care (usually a general ward). This 20 - 30% threshold may be insufficiently stringent for units in severely under-resourced regions, and thresholds may require greater stringency in such units. Conversely, in the privately funded sector, generally greater resource availability means triage thresholds may be considerably less stringent, i.e. patients with similar, or even lower, survival benefit accepted for admission.
In the South African setting, it is recommended that incremental benefit should be largely determined by the likely effect of ICU admission on mortality; however, in some circumstances, when expected quality of life is likely to be severely impaired, even with ICU care, functional outcomes and quality of life should also be considered in decision-making. Examples of such circumstances are provided in Table 3.
As previously stated, to promote consistency in decision-making, and transparency for all stakeholders, individual units are encouraged to develop and document their own thresholds for triage decision- making as far as possible. It would be ideal for thresholds to remain relatively constant over time, but it must be acknowledged that thresholds may change from time to time, depending on periodic changes in resources available and pressure for ICU beds. For example, temporary closure of ICU beds will force a greater number of refusals, and a greater benefit may be required by individual patients to meet admission thresholds. Similar adjustments to the admission threshold may be justified during infectious disease outbreaks. [22,31]
Nevertheless, on a day-to-day basis, a consistent triage threshold based on the above principles is desirable. Consistency of thresholds and decision-making over time allows doctors working within the ICU and referring teams to share appropriate expectations, and promotes fairness for all patients requiring ICU care. This approach specifically does not support the view that the sickest patient should necessarily receive priority, or the view that patients should be admitted only on a first-come, first-served basis. Only in the unusual circumstance when two or more patients under consideration for triage may have an equal triage priority, is admission on a first-referred, first-admitted basis recommended.
4.3. Elective admissions
Postoperative care in ICU is often required for patients who are not currently acutely ill, but require elective surgery and/or have major procedures and/or significant pre-morbid conditions. While delays caused by cancellation may be justified, such surgery can ultimately be considered lifesaving, and of benefit to society at large. Therefore, considerations should be made by individual units to reasonably accommodate elective surgery. These considerations should recognise that ICU length of stay is generally short, and consequently resource use by postoperative cases is relatively small. [32-34] Nevertheless, it must be explicitly acknowledged that, from time to time, resource constraints may result in cancellation/delay of elective cases.
5. Implementation and documentation
5.1. Implementation and responsibility for decision-making
Each ICU in SA that is required to triage patients should have a triage policy. Some barriers to effective implementation of a triage policy can be anticipated; these include lack of acceptance of the triage policy by administrators or healthcare workers, as well as implementation and maintenance costs. Because the additional infrastructure and clinical manpower required to maintain a triage system is small, this should not be a major barrier to implementation. Developing and maintaining a well-accepted policy is more challenging and time consuming. The present guideline is intended to provide a framework on which local triage policy may be modelled. All local policies should be developed and endorsed by a high-level hospital triage committee (or equivalent high-level hospital management committee), chaired by the director of ICU or their nominated representative. Implementation of the local triage policy should also follow a defined administrative process (Table 4) that includes communication and consultation with stakeholders (e.g. ICU doctors, ICU nurses, hospital administrators, potential referring medical teams, and patient advocates). Potential referring teams that should be involved will differ according to the individual unit but would usually include family and emergency medicine, neurosurgery, trauma and general surgery, orthopaedics, obstetrics and gynaecology, general medicine and oncology. The product should demonstrate a rational process that all stakeholders can accept as relevant to fair resource rationing, be fully transparent, and openly published. Lastly, procedures for revising decisions in the light of reasonable challenges to them should be put in place. [35,36] All these aspects should be built into the guideline development process, as was recently described in a South African paediatric ICU setting. [37] Although clinical input from other medical professionals to establish prognosis is valuable, in daily operations the ICU team representative should be ultimately responsible for making decisions on admission and discharge according to this guidance and individual hospital policies. This representative should be a senior ICU doctor, or supervised by a senior ICU doctor. [12,15] In cases of conflict with referring physicians, or the patient, or their surrogate, the ICU director’s decision should be final. [12] It is recommended that the ICU doctor responsible for triage should be the ultimate decision maker, as they are generally the least conflicted by previous association with individual patients, and have the best understanding of expected ICU outcomes, current ICU resource limitations and the resource implications of potential admissions. If there is an irreconcilable disagreement between the ICU director and other clinical departments, the ICU management committee or relevant hospital management committee, including the ethics committee if appropriate, should be responsible for facilitating resolution.
5.2. Monitoring and audit
Triage decisions carry a heavy burden, and ICU refusal on the basis of triage is associated with excess mortality, even after adjustment for severity of illness and comorbidity. [17,38,39] It is therefore important that there should be formal monitoring of the consequences of triage decisions. The guidelines should be reviewed on a regular basis by relevant hospital committees and revised as needed. Performance indicators such as compliance with the guidelines, triage and ICU refusal rates, average length of ICU stay, re-admission rate, and ICU outcomes should also be reviewed regularly, and improvement measures implemented when appropriate. A list of key audit metrics that should be recorded to assist the process of continuous quality improvement can be found in the accompanying consensus statement. [10] Relevant feedback should be clearly communicated to the frontline ICU doctors, hospital administrators as well as relevant regional and national authorities when necessary.
5.3. Documentation
Triage decisions should always be documented in writing in the patient record. This should include the triage priority and clinical reasons for the decision. The decision should be conveyed to the referring doctor/s and, where appropriate, the patient or patient’s surrogate. Transparency and good communication of the reasons for admission or refusal in individual cases is critical to improve all stakeholders’ understanding of the triage process and potentially avoids conflict with other healthcare providers and patients or surrogates.
6. ‘Goals of care’ discussion
In a situation where a patient does not meet the triage threshold for admission, a patient’s or surrogate’s preference for admission must be overridden. This is necessary in order to maintain fairness, and thus triage decisions must be made without patient or surrogate consent. [6] It is, nevertheless, good practice for the ICU doctor to discuss ‘goals of care’ with patients or their surrogates either at the time of ICU referral, or soon after admission to the ICU. [40-43] An honest evaluation and communication to the patient and surrogate of likely prognosis, and the benefits as well as burdens of ICU care, forms an important part of a ‘goal of care’ discussion. Some patients, or surrogates, who have been offered ICU care, may elect to exercise their autonomy at the time of referral by declaring a preference to decline ICU admission if they perceive likely outcome benefits to be outweighed by expected burdens of intensive care and/or the subsequent rehabilitation process. This situation offers patients the ability to exercise their autonomy and make informed medical choices.
While it is desirable to have a discussion with relevant parties establishing a potential ICU patient’s circumstances including functional capacity and quality of life prior to making a triage decision, as it is required to assist decision-making, it is recommended that a formal ‘goals of care’ discussion take place only after the triage decision is finalised and has been communicated to the referring healthcare team and patient or patient’s surrogate. This is important to avoid the circumstance where a patient or surrogate may indicate a desire for admission to ICU when such an option has been denied by the need to triage.
7. Special circumstances
Some patients may be declared dead by formal brain testing. To facilitate the important role of organ donation and the benefits it brings to society, such admissions may be justified in order to ensure the optimal condition of organs for transplantation by facilitating ‘extracranial support’ for a limited period of time. [44] Such ICU admissions are justified on the basis of substantial societal gain for relatively small use of resources.
In some hospitals, alternative facilities such as high-dependency units, or ward ventilation, may be available for patients requiring life support and/or monitoring. While recognising that such care is not optimal, or equivalent to ICU care, some outcome benefit may be achieved for selected individual patients. [45] If no alternatives exist, patients should be offered the best lower level of care available.
Occasionally, patients referred to ICU may have no realistic prospect of deriving benefit from ICU care, and thus ICU admission may be considered ‘futile’ or ‘non-beneficial’. [12,13] A recent consensus conference suggested that the use of the term ‘potentially inappropriate’ be considered unless the treatment requested had no prospect of accomplishing its intended physiological goal, in which case the use of the term ‘futile’ could be considered appropriate. [46] There is a lack of consensus and some controversy surrounding the use of quantitative definitions of non-beneficial interventions (e.g. an intervention that achieves its goal in less than 1 in 100 cases). [6,13,47] The determination of non-beneficial care therefore remains one that should be made by a senior doctor, preferably by consensus with other treating or consulted doctors. Where ICU care is deemed ‘non-beneficial’ or ‘potentially inappropriate’ patients should not be admitted to the ICU. Examples of severely ill or injured patients who might reasonably be considered to fall into this category are provided in Table 5. [12,16] Patients at the other end of the admission spectrum, usually less severely ill or injured, who would derive very little or no anticipated incremental benefit from ICU admission, because equivalent interventions are available in a non-ICU setting, should also be refused admission. Examples may include stable patients after uneventful general anaesthesia for minor limb surgery, or a healthy postpartum mother.
8. Patient discharge from ICU
Ensuring the best use of ICU resources for all patients requires that patients who no longer need intensive care are expeditiously discharged. The status of patients admitted to an ICU should be assessed continuously to identify patients who no longer require ICU care, and can be discharged. General principles to be considered when developing a local discharge guideline follow.
8.1. Routine discharge
Patients who no longer require intensive monitoring or treatment should be discharged immediately. [12,13]
The majority, [48-54] but not all studies, [49,55] have demonstrated that after-hours (night-time) discharge is associated with higher mortality and increased risk for re-admission, [48-54] and after-hours discharge should be avoided unless the bed is immediately required for a new admission.
The care that the patient receives at the discharge destination must be sufficient to provide a safe standard of care appropriate to the patient’s health needs. Therefore, most patients in SA will have to be assessed as stable enough to be suitable for discharge to general wards, whereas some may be reasonably discharged to high- dependency units in a less stable condition if such facilities exist.
Discharge to high-dependency units as a step-down option is likely to be efficient in ‘protecting’ ICU beds for sicker patients who will benefit from ICU admission into freed beds.
8.2. Expedited/early discharge of patients with a good outcome prognosis
Patients who may require additional monitoring, but are not in immediate danger of deterioration without ICU care, may be discharged when a bed is urgently required for another patient with a comparatively higher priority of ICU care. This may be considered when risks to the discharged patient are predicted to be small, and the likely benefit of the patient admitted to the freed bed to be high. Whenever possible, these patients should be discharged to a high- care area.
8.3. Expedited/early discharge of patients with a poor outcome prognosis
Patients whose treatment has failed so that short-term prognosis is poor, or those with little likelihood of recovery and benefit from continued intensive treatment, may be discharged to the ward or other lower levels of care such as high-dependency units for palliative care and/or end-of-life care electively, or when a bed is urgently required for another patient with a comparatively higher priority for ICU care. It is expected that the discharged patient’s prognosis is manifestly very poor, and that the likelihood of benefit to the patient queueing for the freed bed is high. It is recommended that ICUs and other hospital units collaborate with such end-of-life care management, and consider introducing or promoting existing palliative care systems and protocols to assist in the care of such patients.
9. Limitations
The complexity of decision-making and a lack of precise medical knowledge means that a certain amount of inaccuracy will always be present when triage decisions are made. It must be acknowledged that predictions of outcome (e.g. mortality, functional outcomes, and quality of life) and ICU length of stay will, in practice, be imprecise. While some scoring systems are able to accurately predict outcomes such as mortality and ICU length of stay in patient populations, a lack of calibration leads to an inability of models to sufficiently discriminate outcomes accurately in individual patients. [23] In addition, triage decisions must be made within a short period of time, and it is usually not possible to gather all the data required to make score- based predictions in the time frame necessary. Current evidence suggests that scoring systems and clinical calculators are not yet superior to clinical judgment in correctly predicting mortality for individual patients, especially early after presentation. [56] The clinical prediction of mortality by individual doctors, especially when confident about a particular prediction, is relatively good, and rises even better when in concordance with the prediction of others. [57] Nevertheless, prognostic scoring systems, when available, have been recommended to assist and inform a greater degree of quantitative decision-making. [6,13]
In addition to predicting outcome if admitted to the ICU, it is also necessary to predict outcome for critically ill referred patients should they remain in their current care environment, so that incremental benefit can be estimated. Such predictive data for patients outside the ICU are sparse and necessarily rely largely on clinical judgment.
Accurate predictive scores of quality of life after ICU admission for individual patients are similarly unavailable, and the expert group recommends that poor quality of life only be considered as an outcome measure when is likely to be demonstrably and substantially poor (Table 4).
Estimating resource use is also problematic and carries a high degree of uncertainty. Current predictive scores for estimating ICU length of stay (as a surrogate for predicted resource use) have similar problems with predictions for individual patients as for mortality predictions, [58] and models are complex and cannot be readily calculated at the time of admission. [59-61] Available studies suggest that experienced doctors are moderately good at correctly estimating ICU length of stay (LOS). [62] A recent study suggested that while doctors predicted LOS correctly in only about 50% of cases, they under- estimated LOS only in a minority (about 18%) of cases. [63]
Finally, while several of the consensus group responsible for generating the guideline were chosen for their clinical expertise and experience in triage and the practice of intensive care in resource-limited environments, and the guideline was developed after an extensive review of current literature, a limitation of the statement was the lack of additional external expert review during the development process.
10. Conclusion
The process of triage has complex ethical and moral dimensions, and requires clinical expertise to implement effectively and equitably. Triage decisions always require complex judgments and decision- making can be difficult, even for experienced clinicians. These guidelines and the associated decision-making framework are insufficient to solve all the difficulties encountered by ICU doctors and other stakeholders working in SA’s challenging public health resource environment. They do, however, offer an expert consensus of how rationing can be justly applied, and suggest measures that should serve to improve the fairness and consistency with which these decisions are made. The guidelines should also serve as a starting point for further deliberation and/or improvement of triage practices in SA. They may also serve to stimulate research that will
help illuminate the process of decision-making, and help define the magnitude of the resource limitations faced by ICU services in SA. Research is also required to measure relevant outcomes consequent on the practice of triage. The long-term goal is ultimately to allow best delivery of ICU services to those requiring them.
1. Marshall JC, Bosco L, Adhikari NK, et al. What is an intensive care unit? A report of the task force of the World Federation of Societies of Intensive and Critical Care Medicine. J Crit Care 2017;37:270-276. https://doi.org/10.1016/j.jcrc.2016.07.015
2. van Zyl-Smit R, Burch V, Willcox P. The need for appropriate critical care service provision at non-tertiary hospitals in South Africa. S Afr Med J 2007;97(4):268-272.
3. Bhagwanjee S, Scribante J. National audit of critical care resources in South Africa - unit and bed distribution. S Afr Med J 2007;97(12 Pt 3):1311-1314.
4. Naidoo K, Singh J, Lalloo U. Critical analysis of ICU/HC beds in South Africa: 2008-2009. S Afr Med J 2013;103(10):751-753.
5. Skinner DL, De Vasconcellos K, Wise R, et al. Critical care admission of South African (SA) surgical patients: Results of the SA Surgical Outcomes Study. S Afr Med J 2017;107(5):411- 419. https://doi.org/10.7196/SAMJ.2017.v107i5.11455
6. Sprung CL, Danis M, Iapichino G, et al. Triage of intensive care patients: Identifying agreement and controversy. Intensive Care Med 2013;39(11):1916-1924. https://doi.org/10.1007/s00134-013-3033-6
7. Ramos JG, Perondi B, Dias RD, et al. Development of an algorithm to aid triage decisions for intensive care unit admission: A clinical vignette and retrospective cohort study. Crit Care 2016;20:81. https://doi.org/10.1186/s13054-016-1262-0
8. Farmer AP, Legare F, Turcot L, et al. Printed educational materials: Effects on professional practice and health care outcomes. Cochrane Database Syst Rev 2008(3):Cd004398. https://doi.org/10.1002/14651858.CD004398.pub2
9. Kredo T, Bernhardsson S, Machingaidze S, et al. Guide to clinical practice guidelines: The current state of play. Int J Qual Health Care 2016;28(1):122-128. https://doi.org/10.1093/intqhc/mzv115
10. Joynt GM, Gopalan PD, Argent A, et al. The Critical Care Society of Southern Africa Consensus Statement on ICU Triage and Rationing (ConICTri). S Afr Med J 2019;109(8):613- 629. https://doi.org/10.7196.SAMJ.v109i8.13947
11. Lumb P. The Durban declaration. J Crit Care 2013;28(6):887-889. https://doi.org/10.1016/j.jcrc.2013.10.001
12. Guidelines for intensive care unit admission, discharge, and triage. Task Force of the American College of Critical Care Medicine, Society of Critical Care Medicine. Crit Care Med 1999;27(3):633-638.
13. Nates JL, Nunnally M, Kleinpell R, et al. ICU admission, discharge, and triage guidelines: A framework to enhance clinical operations, development of institutional policies, and further research. Crit Care Med 2016;44(8):1553-1602. https://doi.org/10.1097/ccm.0000000000001856
14. Sprung CL, Geber D, Eidelman LA, et al. Evaluation of triage decisions for intensive care admission. Crit Care Med 1999;27(6):1073-1079.
15. Blanch L, Abillama FF, Amin P, et al. Triage decisions for ICU admission: Report from the Task Force of the World Federation of Societies of Intensive and Critical Care Medicine. J Crit Care 2016;36:301-305. https://doi.org/10.1016/j.jcrc.2016.06.014
16. Consensus statement on the triage of critically ill patients. Society of Critical Care Medicine Ethics Committee. JAMA 1994;271(15):1200-1203.
17. Joynt GM, Gomersall CD, Tan P, et al. Prospective evaluation of patients refused admission to an intensive care unit: triage, futility and outcome. Intensive Care Med 2001;27(9):1459-1465. https://doi.org/10.1007/s001340101041
18. Joynt GM, Gomersall CD. Making moral decisions when resources are limited - an approach to triage in ICU patients with respiratory failure. S Afr J Crit Care 2005;21(1):34-44.
19. Joynt GM, Gomersall CD. What do “triage” and “informed consent” really mean in practice? Anaesth Intensive Care 2011;39(4):541-544.
20. Joynt GM, Gomersall C. Integrating elective workloads into an emergency setting in the intensive care unit. In: Flaatten H, Moreno RP, Putensen C, Rhodes A, eds. Organisation and Management of Intensive Care. Berlin: MedizinischWissenschaftlicheVerlagsgesellschaft, 2010:53-64.
21. Brouwers MC, Kho ME, Browman GP, et al. AGREE II: Advancing guideline development, reporting and evaluation in healthcare. CMAJ 2010;182(18):E839-E842. https://doi.org/10.1503/cmaj.090449
22. Cheung WK, Myburgh J, Seppelt IM, et al. A multicentre evaluation of two intensive care unit triage protocols for use in an influenza pandemic. Med J Aust 2012;197(3):178-181.
23. Gomersall CD, Joynt GM. What is the benefit in triage? Crit Care Med 2011;39(4):911-912. https://doi.org/10.1097/CCM.0b013e31820b7415
24. Guest T, Tantam G, Donlin N, et al. An observational cohort study of triage for critical care provision during pandemic influenza: ‘Clipboard physicians’ or ‘evidenced based medicine’? Anaesthesia 2009;64(11):1199-1206. https://doi.org/10.1111/j.1365-2044.2009.06084.x
25. Khan Z, Hulme J, Sherwood N. An assessment of the validity of SOFA score based triage in H1N1 critically ill patients during an influenza pandemic. Anaesthesia 2009;64(12):1283- 1288. https://doi.org/10.1111/j.1365-2044.2009.06135.x
26. Shahpori R, Stelfox HT, Doig CJ, Boiteau PJ, Zygun DA. Sequential organ failure assessment in H1N1 pandemic planning. Crit Care Med 2011;39(4):827-832. https://doi.org/10.1097/CCM.0b013e318206d548
27. Higgins TL, Teres D, Copes WS, et al. Assessing contemporary intensive care unit outcome: An updated mortality probability admission model (MPM0-III). Crit Care Med 2007;35(3):827-835. https://doi.org/10.1097/01.Ccm.0000257337.63529.9f
28. Cowen ME, Czerwinski JL, Posa PJ, et al. Implementation of a mortality prediction rule for real-time decision making: feasibility and validity. J Hosp Med 2014;9(11):720-726. https://doi.org/10.1002/jhm.2250
29. Christian MD, Hawryluck L, Wax RS, et al. Development of a triage protocol for critical care during an influenza pandemic. CMAJ 2006;175(11):1377-1381. https://doi.org/10.1503/cmaj.060911
30. Christian MD, Joynt GM, Hick JL, et al. Chapter 7. Critical care triage. Intensive Care Med 2010;36(Suppl 1):S55-S64. https://doi.org/10.1007/s00134-010-1765-0
31. Christian MD, Fowler R, Muller MP, et al. Critical care resource allocation: Trying to PREEDICCT outcomes without a crystal ball. Crit Care 2013;17(1):107. https://doi.org/10.1186/cc11842
32. Lupei MI, Chipman JG, Beilman GJ, Oancea SC, Konia MR. The association between ASA status and other risk stratification models on postoperative intensive care unit outcomes. AnesthAnalg 2014;118(5):989-994. https://doi.org/10.1213/ane.0000000000000187
33. Joliat GR, Labgaa I, Petermann D, et al. Cost-benefit analysis of an enhanced recovery protocol for pancreaticoduodenectomy. Br J Surg 2015;102(13):1676-1683. https://doi.org/10.1002/bjs.9957
34. Li M, Zhang J, Gan TJ, et al. Enhanced recovery after surgery pathway for patients undergoing cardiac surgery: A randomized clinical trial. Eur J Cardiothorac Surg 2018;54(3):491-497. https://doi.org/10.1093/ejcts/ezy100
35. Daniels N, Sabin J. Limits to health care: Fair procedures, democratic deliberation, and the legitimacy problem for insurers. Philos Public Aff 1997;26(4):303-350.
36. Daniels N. Accountability for reasonableness. BMJ 2000;321(7272):1300-1301.
37. Argent AC, Ahrens J, Morrow BM, et al. Pediatric intensive care in South Africa: An account of making optimum use of limited resources at the Red Cross War Memorial Children’s Hospital. Pediatr Crit Care Med 2014;15(1):7-14. https://doi.org/10.1097/pcc.0000000000000029
38. Simchen E, Sprung CL, Galai N, et al. Survival of critically ill patients hospitalized in and out of intensive care units under paucity of intensive care unit beds. Crit Care Med 2004;32(8):1654-1661.
39. Edbrooke DL, Minelli C, Mills GH, et al. Implications of ICU triage decisions on patient mortality: A cost-effectiveness analysis. Crit Care 2011;15(1):R56. https://doi.org/10.1186/cc10029
40. Bernacki RE, Block SD. Communication about serious illness care goals: A review and synthesis of best practices. JAMA Intern Med 2014;174(12):1994-2003. https://doi.org/10.1001/jamainternmed.2014.5271
41. Joynt GM, Lipman J, Hartog C, et al. The Durban World Congress Ethics Round Table IV: Health care professional end-of-life decision making. J Crit Care 2015;30(2):224-230. https://doi.org/10.1016/j.jcrc.2014.10.011
42. Brighton LJ, Bristowe K. Communication in palliative care: talking about the end of life, before the end of life. Postgrad Med J 2016;92(1090):466-470. https://doi.org/10.1136/postgradmedj-2015-133368
43. Thomas RL, Zubair MY, Hayes B, Ashby MA. Goals of care: A clinical framework for limitation of medical treatment. Med J Aust 2014;201(8):452-455.
44. Cooper DK, De Villiers JC, Smith LS, et al. Medical, legal and administrative aspects of cadaveric organ donation in the RSA. S Afr Med J 1982;62(25):933-938.
45. Tang WM, Tong CK, Yu WC, Tong KL, Buckley TA. Outcome of adult critically ill patients mechanically ventilated on general medical wards. Hong Kong Med J 2012;18(4):284-290.
46. Bosslet GT, Pope TM, Rubenfeld GD, et al. An official ATS/AACN/ACCP/ESICM/SCCM policy statement: responding to requests for potentially inappropriate treatments in intensive care units. Am J Respir Crit Care Med 2015;191(11):1318-1330. https://doi.org/10.1164/rccm.201505-0924ST
47. Schneiderman LJ, Jecker NS, Jonsen AR. Medical futility: Its meaning and ethical implications. Ann Intern Med 1990;112(12):949-954.
48. Duke GJ, Green JV, Briedis JH. Night-shift discharge from intensive care unit increases the mortality-risk of ICU survivors. Anaesth Intensive Care 2004;32(5):697-701.
49. Hanane T, Keegan MT, Seferian EG, Gajic O, Afessa B. The association between nighttime transfer from the intensive care unit and patient outcome. Crit Care Med 2008;36(8):2232-2237. https://doi.org/10.1097/CCM.0b013e3181809ca9
50. Laupland KB, Shahpori R, Kirkpatrick AW, Stelfox HT. Hospital mortality among adults admitted to and discharged from intensive care on weekends and evenings. J Crit Care 2008;23(3):317-324. https://doi.org/10.1016/j.jcrc.2007.09.001
51. Laupland KB, Misset B, Souweine B, et al. Mortality associated with timing of admission to and discharge from ICU: A retrospective cohort study. BMC Health Serv Res 2011;11:321. https://doi.org/10.1186/1472-6963-11-321
52. Tobin AE, Santamaria JD. After-hours discharges from intensive care are associated with increased mortality. Med J Aust 2006;184(7):334-337.
53. Singh MY, Nayyar V, Clark PT, Kim C. Does after-hours discharge of ICU patients influence outcome? Crit Care Resusc 2010;12(3):156-161.
54. Ouanes I, Schwebel C, Francais A, et al. A model to predict short-term death or readmission after intensive care unit discharge. J Crit Care 2012;27(4):422.e421-429. https://doi.org/10.1016/j.jcrc.2011.08.003
55. Makris N, Dulhunty JM, Paratz JD, Bandeshe H, Gowardman JR. Unplanned early readmission to the intensive care unit: A case-control study of patient, intensive care and ward-related factors. Anaesth Intensive Care 2010;38(4):723-731.
56. Sinuff T, Adhikari NK, Cook DJ, et al. Mortality predictions in the intensive care unit: Comparing physicians with scoring systems. Crit Care Med 2006;34(3):878-885. https://doi.org/10.1097/01.Ccm.0000201881.58644.41
57. Detsky ME, Harhay MO, Bayard DF, et al. Discriminative accuracy of physician and nurse predictions for survival and functional outcomes 6 months after an ICU admission. JAMA 2017;317(21):2187-2195. https://doi.org/10.1001/jama.2017.4078
58. Woods AW, MacKirdy FN, Livingston BM, Norrie J, Howie JC. Evaluation of predicted and actual length of stay in 22 Scottish intensive care units using the APACHE III system. Acute physiology and chronic health evaluation. Anaesthesia 2000;55(11):1058-1065.
59. Zimmerman JE, Kramer AA, McNair DS, Malila FM, Shaffer VL. Intensive care unit length of stay: Benchmarking based on Acute Physiology and Chronic Health Evaluation (APACHE) IV. Crit Care Med 2006;34(10):2517-2529. https://doi.org/10.1097/01.Ccm.0000240233.01711.D9
60. Verburg IW, Atashi A, Eslami S, et al. Which models can I use to predict adult ICU length of stay? A systematic review. Crit Care Med 2017;45(2):e222-e231. https://doi.org/10.1097/ccm.0000000000002054
61. Kramer AA. Are ICU length of stay predictions worthwhile? Crit Care Med 2017;45(2):379- 380. https://doi.org/10.1097/ccm.0000000000002111
62. Gusmao Vicente F, Polito Lomar F, Melot C, Vincent JL. Can the experienced ICU physician predict ICU length of stay and outcome better than less experienced colleagues? Intensive Care Med 2004;30(4):655-659. https://doi.org/10.1007/s00134-003-2139-7
63. Nassar AP, Jr., Caruso P. ICU physicians are unable to accurately predict length of stay at admission: A prospective study. Int J Qual Health Care 2016;28(1):99-103. https://doi.org/10.1093/intqhc/mzv112
Article Views
Full text views: 3791




1.jpg)
Comments on this article
*Read our policy for posting comments here